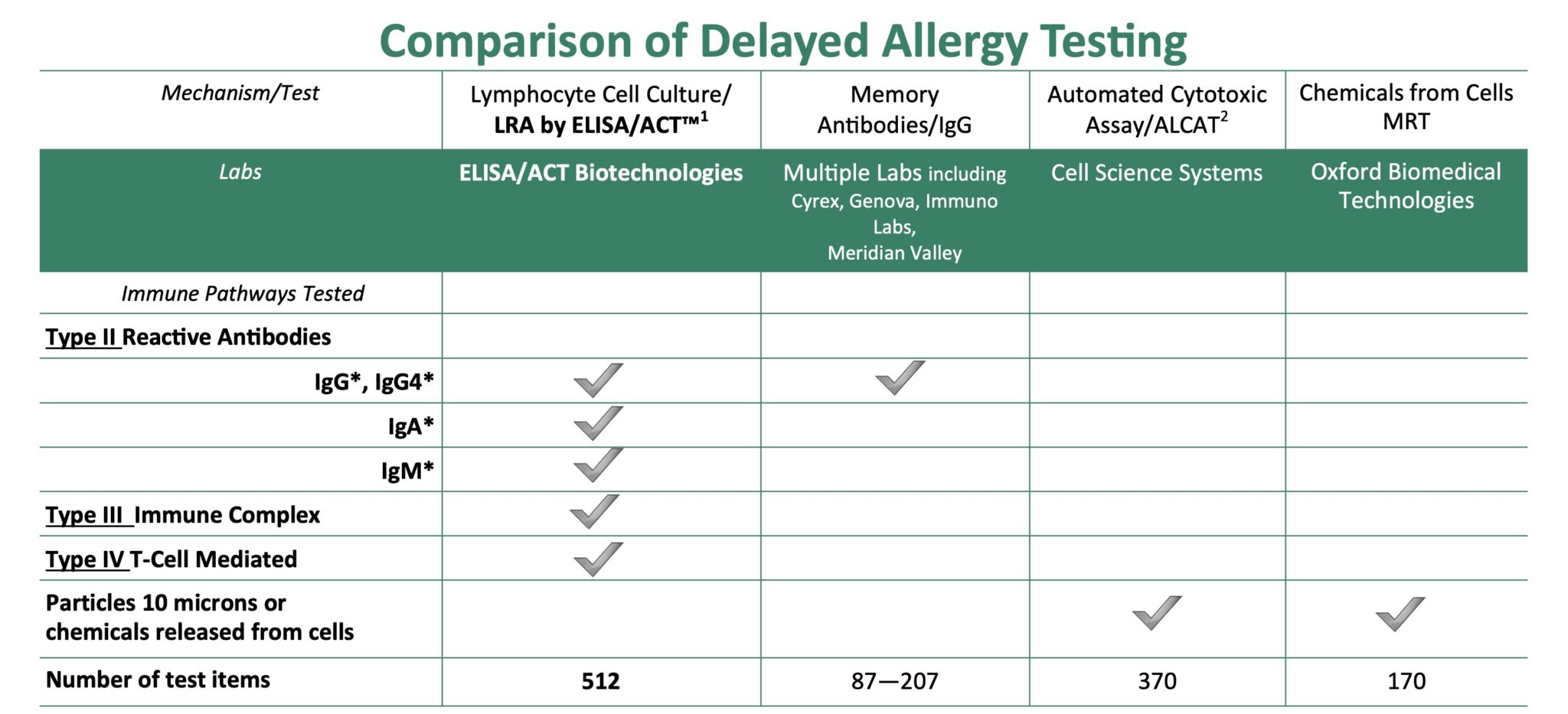
Lab Test Comparison
Only LRA directly measures lymphocyte response. By directly monitoring lymphocytes, LRA tests...
- Identify ONLY true reactive items (less than .1% false positives)
- Can detect ALL 3 types of delayed hypersensitivity
- Are HIGHLY reproducible (less than 3% day to day variance)
The table above compares LRA testing with other common tests of delayed hypersensitivity. See links below for additional information.
Read about LRA vs IgG testing for gluten sensitivity.
Study concludes poor reproducibility of cell size testing.
1Only ELISA/ACT Biotechnologies LLC’s (EAB) Lymphocyte Response Assay (LRA) by ELISA/ACT® measures all three types of delayed sensitivity reactions through activation of lymphocytes just as they occur in the body.
2Measures a composite of granulocyte destruction, activation of blood clotting, and complement activation of white cells. Not a functional immunology procedure. Does not measure lymphocyte-specific responses.
* Only a functional, cell-based assay can distinguish between protective (“good”; non-complement activating) or reactive (“bad”; complement activating) antibodies since lymphocyte cells are turned on by clinically active antibodies. Protective antibodies mean people are tolerant (“immunized”) and may eat those substances. Measurement of total antibodies is not related to hypersensitivity. Only antigen-specific measures have clinical meaning.